Home | Introduction | Oxygen Binding | F Helix Transition | Quarternary Shift | Synopsis | Bibliography

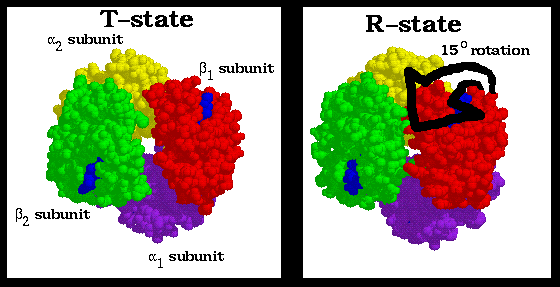
Because of the rigid nature of the alpha1-beta1 and alpha2-beta2 interfaces, the reorientation at the alpha1-beta2 interface can only occur with a corresponding reorientation at the alpha2-beta1 interface. Thus oxygen binding at any of the heme groups leads to a quarternary shift involving both alpha1-beta1 and alpha2-beta2 protomers. This results in a 15 degree rotation of the protomers with respect to each other (shown above, the red and purple alpha1-beta1 protomer is rotating with respect to the green and yellow alpha2-beta2 protomer). During this rotation, the alpha1-beta2 and alpha2-beta1 interfaces act as a flexible joint that the protein uses to alternate between the T and R states.
Say, for example, a oxygen binds to the heme group on the alpha1 subunit. This causes the previously mentioned shift at the alpha1-beta2 interface. Because of the inflexible nature of the alpha1-beta1 interface, there is a corresponding shift at the beta1-alpha2 interface. These shifts require rearrangments of residues in each of the subunits. Ultimately these rearrangments cause all of the heme groups to be flattened into their planar R-state form. Note that no matter which heme group the first oxygen binds to, the result is that all heme groups become flattened.
This flattening has important implications. The domed heme of the T-state Hb creates a strain on the oxygen-iron(II) bond because of steric interactions between the porphyrin ring and the oxygen. This strain is relaxed in the R-state by the flattening of the heme plane of all the heme groups, making it easier for future oxygen binding events to occur. Thus a result of the quarternary shift of Hb from the T-state to the R-state is increased oxygen binding affinity.